Loading market data...
Cipla's U.S. launch of generic Saxenda (liraglutide) signals a major play in the global weight management market. FinScann analyzes the financial impact and implications for Indian pharma and investors.
Pharmaceutical major Cipla Ltd. has announced the immediate launch of Liraglutide Injection, 18 mg/3 mL (6 mg/mL) single-patient-use prefilled pens, the generic equivalent of Saxenda® (liraglutide injection), in the United States. This significant development, executed through its wholly-owned subsidiary Cipla USA Inc. and partner Orbicular, positions Cipla as a key player in the burgeoning global weight management and anti-obesity drug market. While this initial launch targets the U.S. market, it carries profound implications for the rapidly expanding Indian weight management sector, which is projected to reach ₹546.70 million (USD 546.70 million) by FY2033 for anti-obesity drugs, growing at a robust CAGR of 23.54% from FY2026. The move underscores Cipla's strategic intent to capture a share in the high-demand GLP-1 agonist segment, offering potentially more affordable solutions to a patient population grappling with rising obesity rates.
The Catalyst
The immediate catalyst for this announcement is the U.S. Food & Drug Administration (USFDA) Abbreviated New Drug Approval (ANDA) obtained by Cipla's partner, Orbicular, for Liraglutide Injection. Cipla will spearhead the marketing and distribution efforts in the U.S., ensuring broad access and reliable availability for patients. This launch is particularly timely given the immense global and domestic demand for effective weight loss solutions. The active ingredient, liraglutide, is a glucagon-like peptide-1 (GLP-1) receptor agonist, a class of drugs that has revolutionized obesity treatment.
While the U.S. market is the initial focus, the broader context points towards significant opportunities in India. The patent for semaglutide, another prominent GLP-1 agonist found in drugs like Wegovy and Ozempic, is set to expire in India in 2026. This has led major Indian pharmaceutical players, including Cipla, Dr. Reddy's, Sun Pharmaceutical Industries, and Zydus, to actively anticipate and prepare for the end of Novo Nordisk's exclusivity on these crucial GLP-1 medications. The Indian government has also expressed plans to subsidize the manufacturing of diabetes and weight-loss treatments following semaglutide's patent expiration, further signaling a supportive environment for generic competition.
Financial Forensics
Cipla's entry into the generic Saxenda market could significantly bolster its revenue streams, especially as the company navigates recent challenges. In February 2026, Cipla's shares experienced downward pressure following the US FDA's "Official Action Indicated" (OAI) classification for a manufacturing facility in Greece, impacting its supply partner for Lanreotide Injection. This regulatory hurdle and the phasing out of generic Revlimid have led to revised margin guidance and projected weaker performance in Q4 FY26.
However, Cipla has demonstrated resilience, reporting a 30% year-on-year (YoY) increase in net profit to ₹1,222 crore for Q4 FY25, with revenue rising 8.45% YoY to ₹6,597.72 crore. The launch of generic liraglutide represents a strategic move to diversify its portfolio and tap into a high-growth therapeutic area. The U.S. GLP-1 weight-management market alone was estimated at approximately USD 127 million as of December 2025.
The Indian weight management market is experiencing exponential growth, driven by increasing obesity rates, rising health consciousness, and an expanding healthcare infrastructure. According to research, the Indian anti-obesity drugs market, valued at USD 80.0 million in 2024, is projected to reach USD 190.0 million by 2033, with a CAGR of 9.9% from 2025-2033. Other estimates project the Indian anti-obesity drugs market to grow from USD 100.76 million in FY2025 to USD 546.70 million in FY2033, demonstrating a remarkable CAGR of 23.54% during this period. This robust growth trajectory presents a substantial opportunity for companies offering affordable generic alternatives.
Comparative Landscape: Innovator vs. Generic Liraglutide
| Feature | Innovator Saxenda (Liraglutide) | Cipla's Generic Liraglutide (U.S. Launch) |
|---|---|---|
| Active Ingredient | Liraglutide | Liraglutide |
| Indication | Chronic weight management (adjunct to diet & exercise) | Chronic weight management (adjunct to diet & exercise) |
| Delivery | Prefilled multi-dose pen | Prefilled multi-dose pen |
| Regulatory Approval | Original FDA approval | USFDA ANDA approval for generic equivalent |
| Market | Global, established (U.S., Europe, etc.) | Initially U.S. |
| Pricing Strategy | Premium pricing | Expected to be more affordable, driving market access |
| Competition | Semaglutide (Wegovy, Ozempic), Tirzepatide (Mounjaro, Zepbound) | Innovator, other generic entrants (e.g., Teva in US) |
| Impact on Access | Limited by high cost for many patients | Significantly enhances affordability and patient access |
| India Market Entry | Novo Nordisk's Saxenda present, Semaglutide patents expiring in 2026 in India | Potential future launch in India, leveraging global expertise |
| FinScann Source: Internal Analysis and |
Market Impact
Cipla's foray into generic liraglutide positions it strategically within the GLP-1 landscape. While liraglutide is considered an earlier generation GLP-1 compared to newer, once-weekly semaglutide or tirzepatide, its established efficacy and the potential for a significantly lower price point make it highly attractive, especially in price-sensitive markets like India.
The entry of generic versions, such as Teva's generic Saxenda in the U.S. in August 2025, has already begun to reshape pricing dynamics. Teva's generic was introduced at a slight reduction from the brand price in the U.S., and the arrival of generic competition can lead to original suppliers losing substantial market share and drug prices decreasing by 20-30%. This competitive pricing will democratize access to these life-changing therapies.
For India, the impact could be even more profound. With nearly 100 million Indians affected by adult obesity and a strong correlation with comorbidities like Type 2 diabetes and cardiovascular disease, the need for affordable and accessible obesity management solutions is critical. Leading global players like Novo Nordisk and Eli Lilly are already vying for market share in India's burgeoning anti-obesity drug market, with products like Ozempic and Mounjaro recently launched. Cipla, a prominent Indian pharmaceutical company, is well-positioned to leverage its robust domestic distribution network and manufacturing capabilities to introduce affordable generic options once regulatory pathways and patent expirations allow for a similar launch in India. This move would align perfectly with the Indian government's push for affordable healthcare.
Loading chart...
Key Takeaways
FinScann Verdict
Cipla's launch of generic Saxenda in the U.S. is a decisive step that reinforces its commitment to high-growth, complex generics. For the Indian market, this signals Cipla's intent to become a formidable player in the domestic weight management segment as GLP-1 patents expire. Investors should view this as a strategic long-term play that could significantly contribute to Cipla's growth trajectory, offering crucial affordable healthcare solutions to millions in India.
Q: What is Saxenda and what is its generic name? A: Saxenda is a brand-name prescription medication used for chronic weight management. Its active ingredient is liraglutide, which is also the generic name. Liraglutide belongs to the class of GLP-1 receptor agonists.
Q: Is Cipla's generic Saxenda available in India? A: As of February 2026, Cipla has announced the launch of its generic liraglutide (equivalent to Saxenda) in the United States through its subsidiary Cipla USA Inc. While the article highlights the strong potential for such a product in India given the burgeoning weight management market and upcoming patent expirations for related GLP-1 drugs, an official launch in India by Cipla has not been announced. Indian drugmakers, including Cipla, are keenly watching GLP-1 patent expirations in India, particularly for semaglutide in 2026.
Q: How big is the anti-obesity drug market in India? A: The Indian anti-obesity drugs market was valued at USD 80.0 million in 2024 and is projected to reach USD 190.0 million by 2033, exhibiting a Compound Annual Growth Rate (CAGR) of 9.9% during this period. Other reports indicate the market could grow from USD 100.76 million in FY2025 to USD 546.70 million in FY2033, with a CAGR of 23.54% from FY2026-FY2033.
Q: What is the significance of GLP-1 drugs in weight management? A: GLP-1 (Glucagon-Like Peptide-1) receptor agonists are a class of medications that mimic a natural hormone, helping to regulate appetite, slow digestion, and improve blood sugar control. They are highly effective in promoting weight loss and are increasingly prescribed for chronic weight management.
Disclaimer: For information only; not investment advice. Stock market investments carry risks. Please consult a SEBI-registered advisor before investing. FinScann assumes no liability for decisions made based on this report.

Financial journalist specializing in market analysis, stock research, and investment trends. Dedicated to providing accurate, timely insights for informed decision-making.
Credentials: Experienced financial journalist with expertise in equity markets and economic analysis
The information provided in this article is for educational and informational purposes only and should not be construed as financial, investment, or legal advice. Finscann does not provide personalized investment recommendations.
For detailed terms and conditions, please read our Disclaimer and Terms of Service.
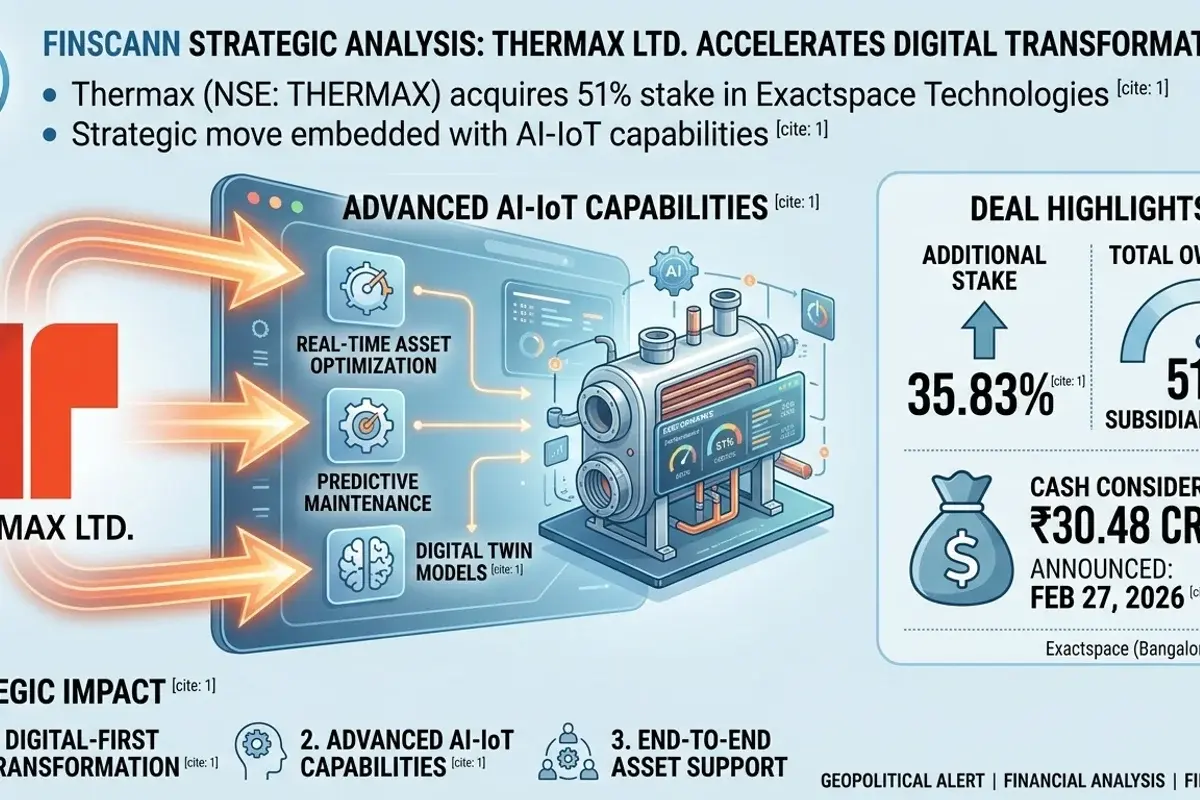
Thermax Ltd. acquires a 51% stake in AI-IoT specialist Exactspace Technologies for ₹30.48 crore, accelerating its digital transformation in India's...

Jio Financial Services launches the AI-powered JioFinance app in February 2026, transforming India's digital finance with personalized loans,...

Tata Steel has completed the sale of its Jajpur Ferro Alloy Plant for ₹610 crore to IMFA, aligning with its portfolio optimization strategy.

IIFL Home Finance secures a landmark $300M ADB loan, its first syndicated external commercial borrowing, enhancing affordable housing access for...

Aditya Birla Capital (ABCAPITAL) subsidiary, Aditya Birla Sun Life Insurance, secures a coveted AAA rating for its ₹1,500 Cr subordinate debt,...